

A new approach reveals unexpected ways oxygen can bind to rare metals.
Iron plays a central role in how the body uses oxygen. In hemoglobin, it binds dioxygen, a pair of oxygen atoms, allowing blood to carry oxygen to tissues. But this is only part of the story. Iron-oxo compounds, which contain iron bonded to oxygen in a highly reactive form, also drive critical chemistry in the liver, where enzymes rely on them to break down medications and toxins.
Rice University chemist Raúl Hernández Sánchez set out to explore whether oxygen could react with other metals, particularly those in the lowest region of the periodic table known as the f-block. This group includes lanthanides in the upper row and actinides below.
He proposed that if lanthanides could bond with oxygen, they might form reactive lanthanide-oxo compounds. These compounds could serve as synthetic alternatives to iron-oxo systems and give chemists new ways to study small-molecule reactions linked to biology.
A Fundamental Limitation
One major obstacle is that f-block metals, especially lanthanides, typically cannot interact with small molecules like oxygen through pi interactions. These interactions are critical in many biological systems, including proteins. In a study published in the Journal of the American Chemical Society, Hernández Sánchez and his team reported a way to overcome this limitation. They showed how to enable pi interactions between dioxygen and a lanthanide metal called neodymium, allowing lanthanide-oxo compounds to form.
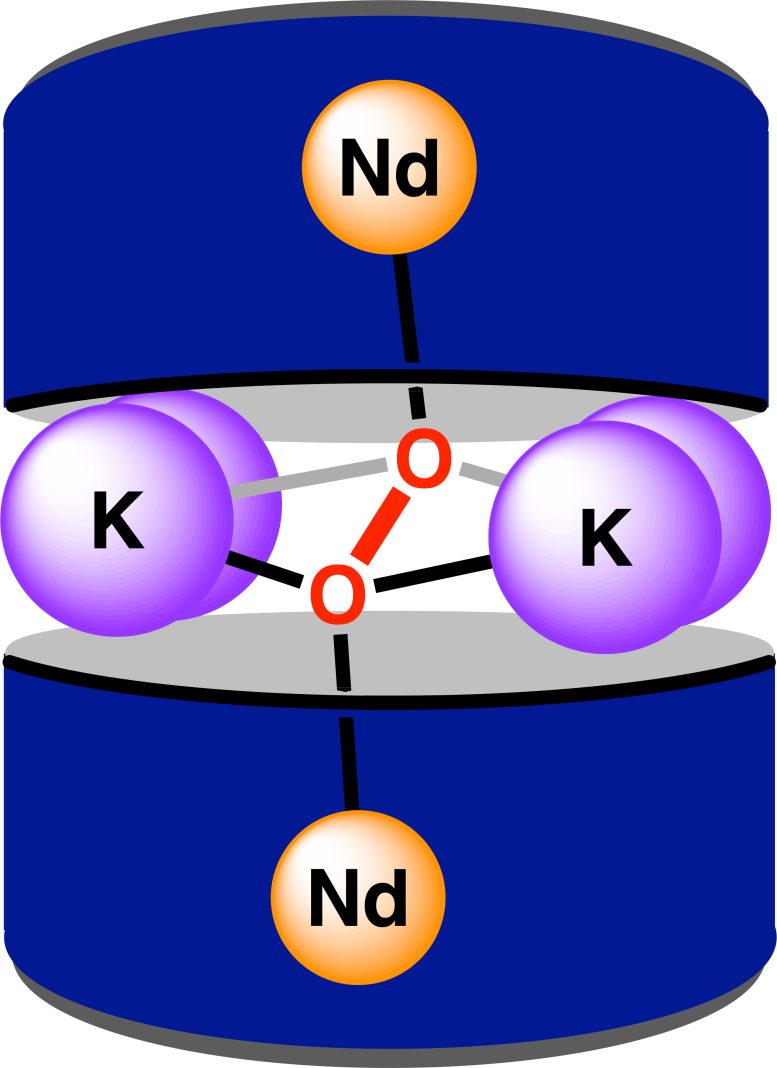
“We had a ligand platform that we developed a few years ago,” said Hernández Sánchez, an assistant professor of chemistry. “You can think of it as a basket that allows us to capture metals and position them in ways to encourage specific types of bindings.”
This “basket” is designed to hold a single f-block metal atom. The researchers arranged two of these structures opposite each other and placed six carefully positioned atoms between them, including a dioxygen molecule that connects the two neodymium centers. This setup creates an octacoordinate ligand environment, which allows precise control over how the metals are positioned.
Achieving an Unprecedented Interaction
“Once we had the lanthanide in our ligand basket, we started to explore its reactivity to small molecule substrates until we found the right conditions to find dioxygen in an unprecedented fashion,” said Hong-Lei Xu, a postdoctoral researcher and first author on the paper.
Under these conditions, which go against earlier assumptions, pi interactions between neodymium and dioxygen became possible. This led to the formation of a lanthanide-oxo molecule. Researchers can now investigate whether these highly reactive compounds can replace iron-oxo systems and explore what new capabilities they might offer.
Although the study focused on neodymium, the team believes the same strategy could apply to many lanthanides and possibly actinides using the same ligand framework.
“The ability to bind dioxygen to f-block metals and cleave the bond between the two oxygen atoms allows us to potentially unveil highly reactive lanthanide oxos and form high value-added chemicals. We could open a new chapter in the chemistry of lanthanides,” Hernández Sánchez said.
Reference: “Activation of Dioxygen via Neodymium-Alkali Metal Clusters” by Hong-Lei Xu, Alejandro Fuentes Beltrán and Raúl Hernández Sánchez, 20 March 2026, Journal of the American Chemical Society.
DOI: 10.1021/jacs.5c22234
This research was supported with startup funds provided by Rice University, the Robert A. Welch Foundation and a Welch Foundation Grant (C-2142-20230405).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.




